Science online The electronic configuration and the chemical activity
Hydrogen. Helium. Lithium. Oxygen. Carbon. Nitrogen. Neon. Magnesium. Silicon. Sulfur. Iron. Aluminum. Calcium. Argon. Sodium. Krypton. Xenon. Barium. Strontium. JPEG screen grabs of an applet which computes and plots the spectra in a web browser window. The above images aren't dithered. They may appear so if your display doesn't have enough.
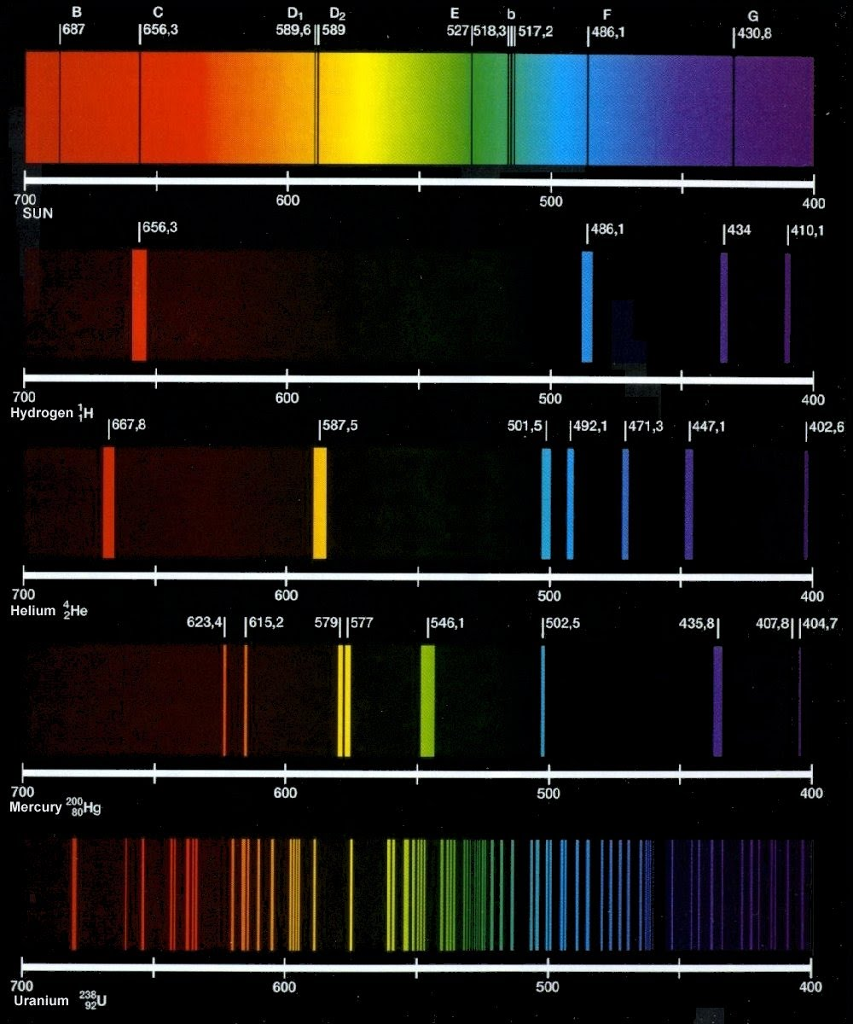
Solved Di D2 589,6 1589 656,3 527518311517,2 1486,1 430,8
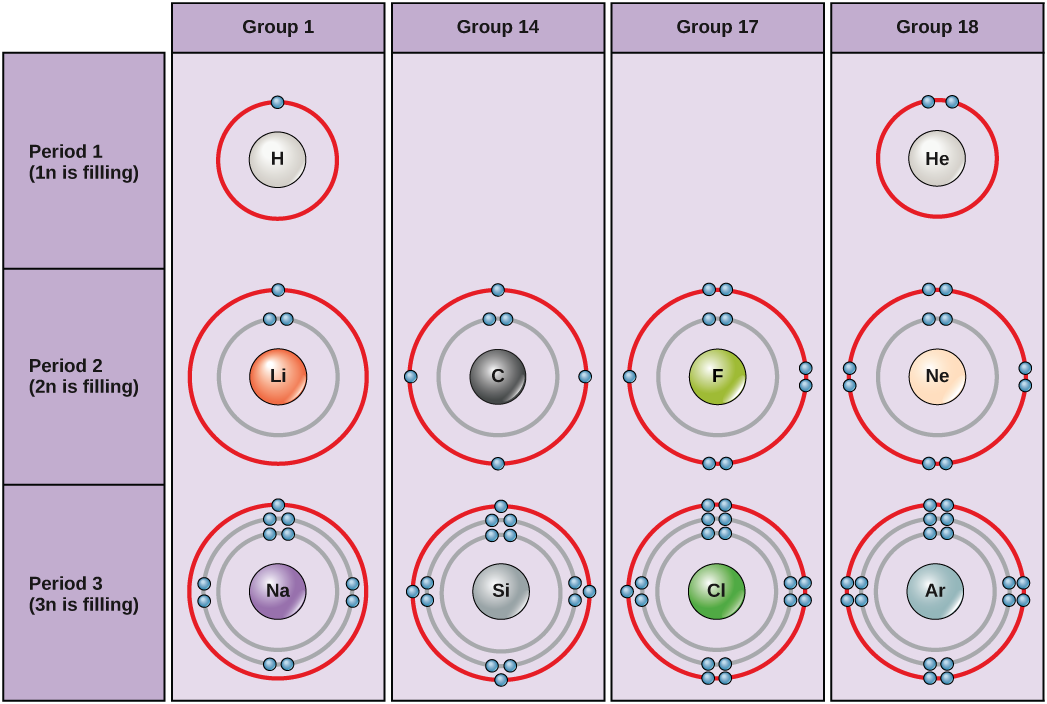
Bohr diagrams for hydrogen, helium, lithium, carbon, fluorine, neon, sodium, silicon, chlorine, and argon. Bohr diagrams indicate how many electrons fill each principal shell. Group 18 elements (helium, neon, and argon are shown in Figure 2) have a full outer, or valence, shell. A full valence shell is the most stable electron configuration.
why were hydrogen and helium the first elements created Michael Dertuary1989
Electronegativity is a chemical property which describes how well an atom can attract an electron to itself. Values for electronegativity run from 0 to 4. Electronegativity is used to predict whether a bond between atoms will be ionic or covalent.

Hydrogen, Helium, Lithium Loops Set Stock Motion Graphics Motion Array
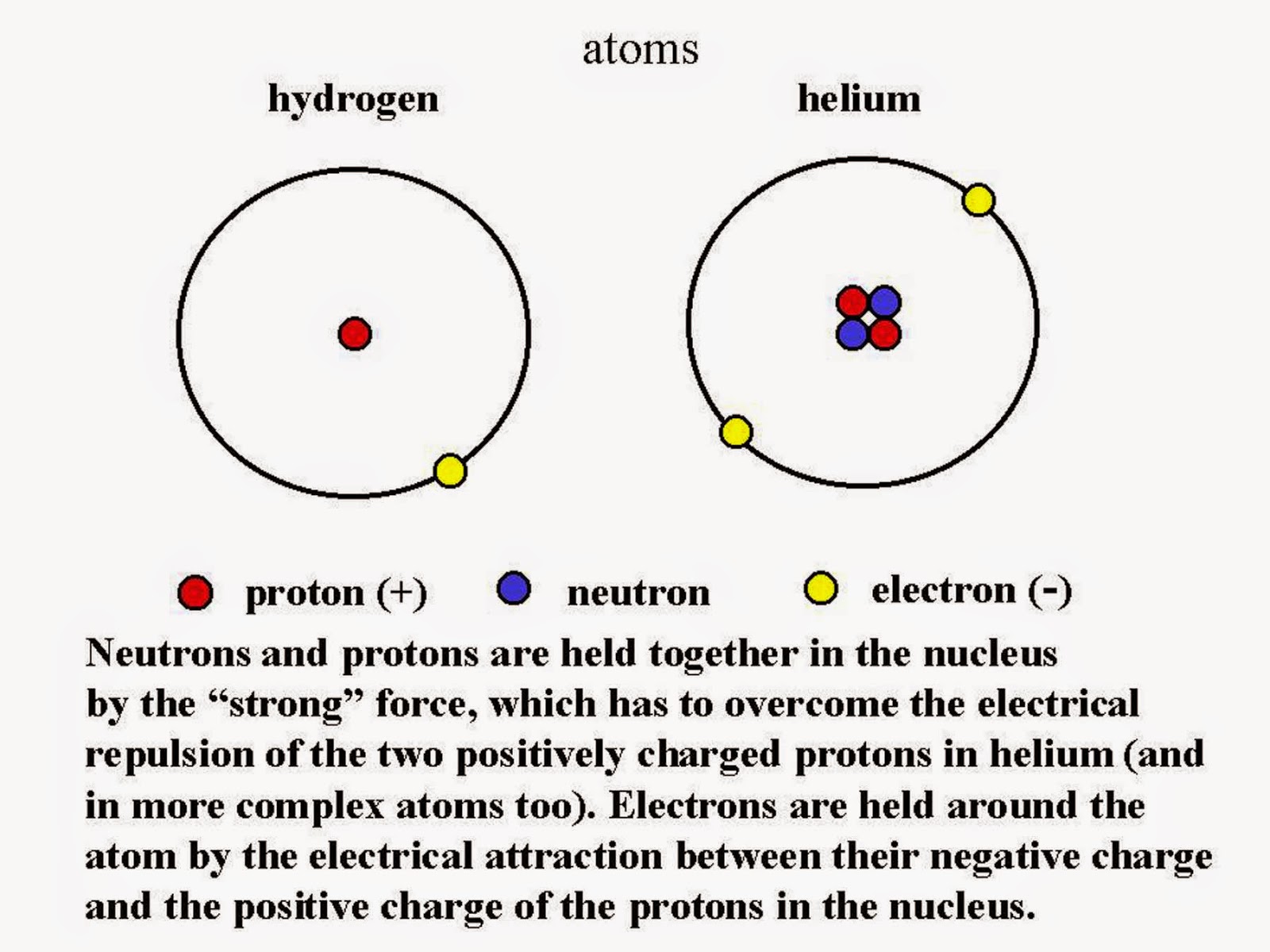
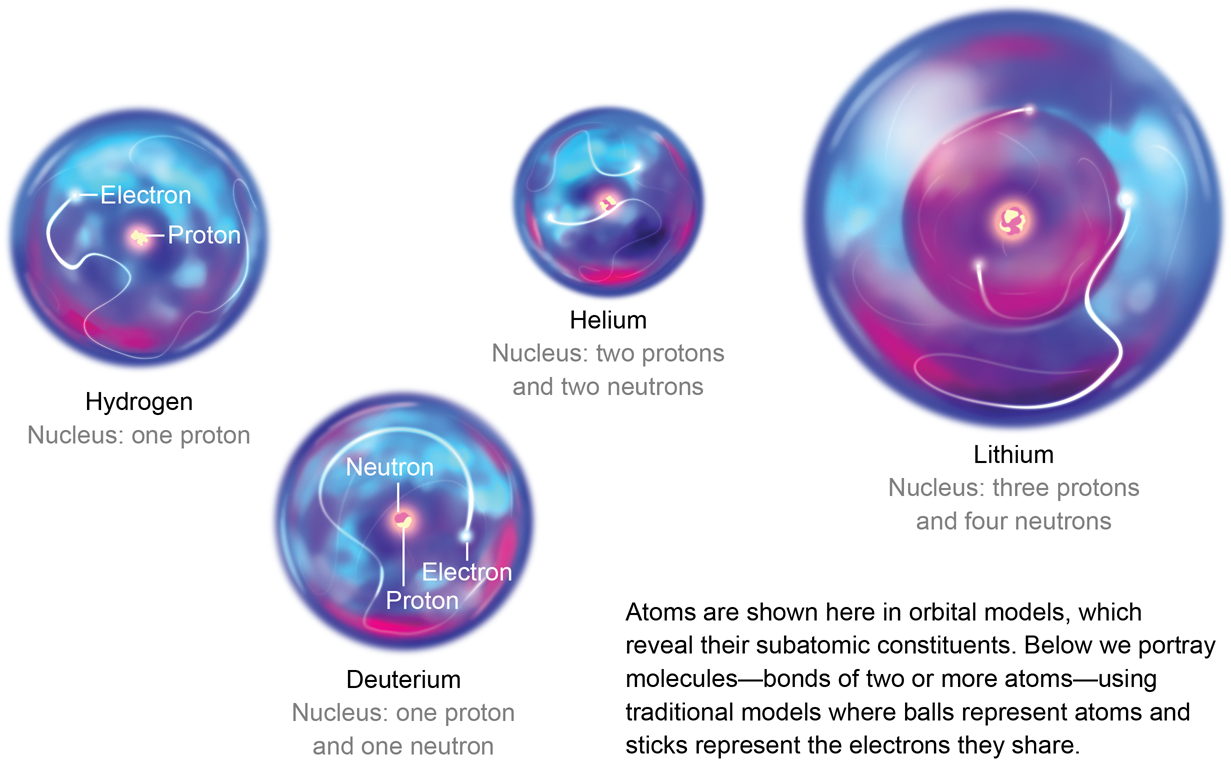
The Bohr model shows the atom as a central nucleus containing protons and neutrons, with the electrons in circular electron shells at specific distances from the nucleus, similar to planets orbiting around the sun.

1 H Hydrogen 2 He Helium 3 Li Lithium 4 Be
Hydrogen is the most abundant element in the Universe; helium is second. However, after this, the rank of abundance does not continue to correspond to the atomic number; oxygen has abundance rank 3, but atomic number 8. All others are substantially less common.

Table Périodique D'éléments Fondamentaux Illustration Stock Illustration du azote, actif 6527575
5.14: Hydrogen, Helium, Lithium. With some familiarity with the properties of single electrons, such as the single electron around the hydrogen nucleus above, we can discuss atoms containing more than one electron. The images found here depict electron wave density by number of dots. Thus, more dots indicates more electron density 'cloud' in.
Elektronen Biologie für Hauptfach I Great Journey
Lithium is rare in the Universe, although it was one of the three elements, along with hydrogen and helium, to be created in the Big Bang. The element was discovered on Earth in 1817 by Johan August Arfvedson (1792-1841) in Stockholm when he investigated petalite, one of the first lithium minerals to be discovered.
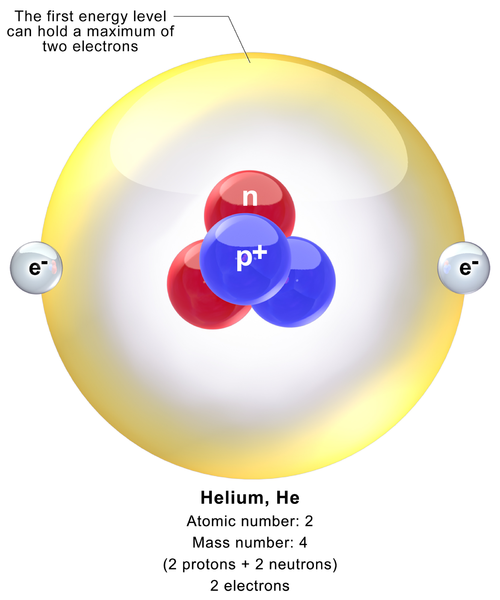
Difference Between Helium and Hydrogen Properties, Isotopes, Reactions, Applications
The periodic table is a chart that organizes the elements by their atomic number. The first 30 elements are hydrogen, helium, lithium, beryllium, boron, carbon, nitrogen, oxygen, fluorine, neon, sodium, magnesium, aluminum, silicon, and phosphorus.

hydrogen helium lithium beryllium... Drawception
nuclear astrophysics High-energy nuclear physics Scientists Physics portal Category v t e Nuclear fusion is a reaction in which two or more atomic nuclei, usually deuterium and tritium (hydrogen variants), combine to form one or more different atomic nuclei and subatomic particles ( neutrons or protons ).

Unique Periodic Table song Hydrogen Helium Lithium Beryllium tablepriodic priodic
Hydrogen has no neutrons, helium has two, lithium has four and beryllium has five, and the masses of the elements increase in that order. Hydrogen and helium are gases, whereas lithium and beryllium are metals. The Periodic Table and the Masses of Elements You can easily identify the lightest elements by checking the periodic table (see Resources).

Hydrogen and Lithium Comparison Properties Material Properties
The atomic number of an element provides insight into the number of protons that exist inside the nuclei of the atoms of that element and also into the number of electrons that surround these nuclei. For example, the atomic number of sodium is 11.

Periodic Table Hydrogen Periodic Table Timeline
Figure 6.19.1 6.19. 1 The figures above show the electron density of different elements. On the left from top to bottom are Hydrogen, Helium, and Lithium. On the right from top to bottom are Beryllium, Boron, and Carbon. Hydrogen has a larger circular area concentrated with dots when compared to helium.
Why does hydrogen naturally form diatomic molecules (H2), whereas helium exists as a single atom
Therefore, only hydrogen, helium and lithium were available when the earliest stars formed several hundred million years later. As we have seen, almost all of this material was 1 H and 4 He. The reactions that could form the lightest elements beyond hydrogen and helium are shown in Figure 16.8. The formation of beryllium is speculative, as we.

Atomic structure of Hydrogen, Helium,Lithium,Beryllium,Boron,Carbon,Nitrogen,Oxygen,Fluorine
Li = 75:62eV for helium and lithium, respec-tively. For the ionization of the third lithium electron an energy of I(3) Li = 122:42eV is required. For full thermal ionization of all helium or lithium electrons we need far more than 100eV, i.e. more than 106 K. An alternative way to reach high ionization degrees is to increase the particle
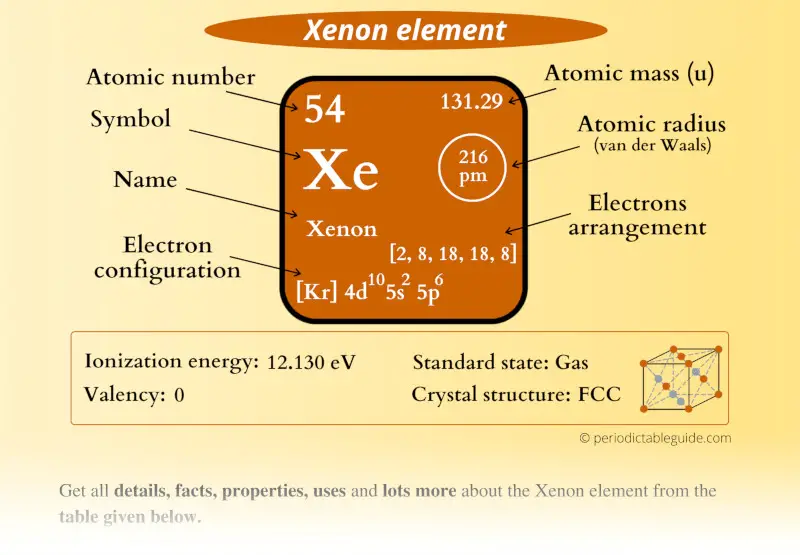
Xenon (Xe) Periodic Table (Element Information & More)
Example 1: Helium vs. Lithium. Hydrogen has an electronic structure of 1s 1. It is a very small atom, and the single electron is close to the nucleus and therefore strongly attracted. There are no electrons screening it from the nucleus and so the ionization energy is high (1310 kJ mol-1). Helium has a structure 1s 2. The electron is being.

Hydrogen (H) Helium (He) Lithium (Li) Beryllium (Be) Boron (B DocsLib
You may assume that the valences of the elements—the number of electrons with which an atom will bond or form—are those that can be derived by looking at the groups (columns) of the periodic table. While these are the most common valences, the real behavior of electrons is less simple. Here is a table of element valences.